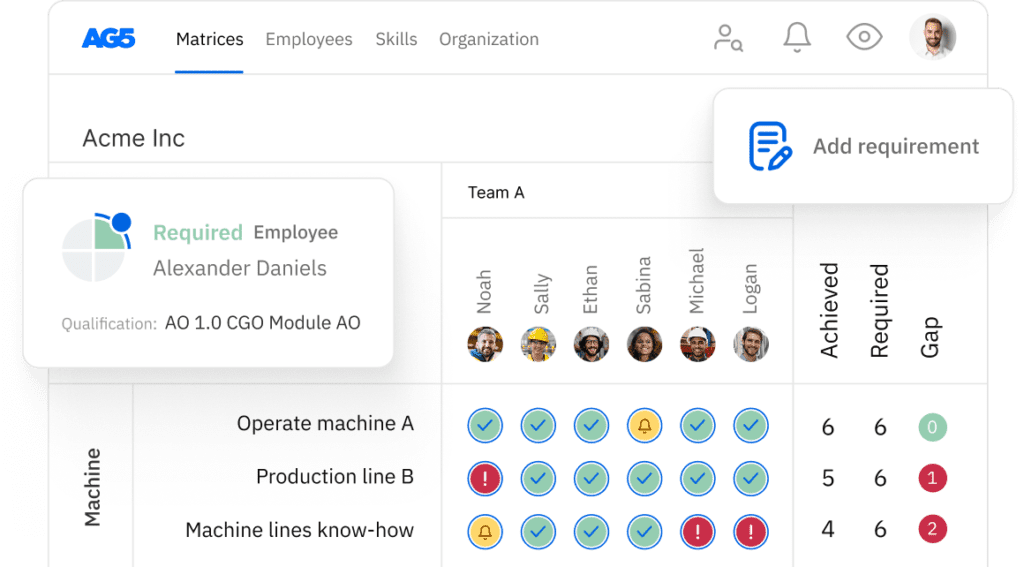
Clinical trial coordination skills matrix template
A skills matrix template is a tool teams can use to assess their clinical trial coordination skills and knowledge
Download your free template here
Overview Copied
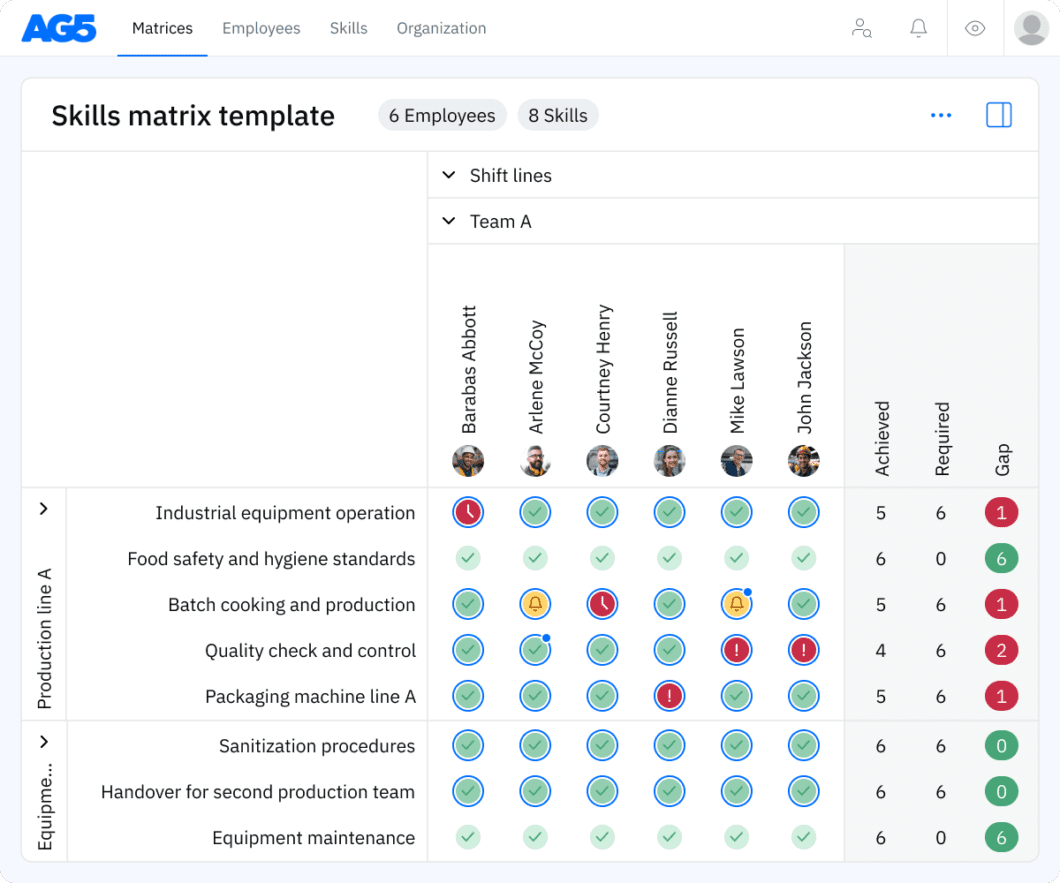
With our free clinical trial coordination skills matrix template, you will receive a clear overview of the skills that are present in your organization, as well as those that are missing. Using this information, you can develop and implement a plan to ensure that your employees’ skills are up to date, comprehensive, compliant, and ready for the future.
Supply chain management for clinical trials
- Managing the procurement and distribution of investigational products
- Coordinating the supply of trial materials with manufacturing timelines
- Ensuring compliance with Good Manufacturing Practices (GMP) in the supply chain
- Monitoring inventory levels and expiration dates of trial materials
- Implementing cold chain logistics for temperature-sensitive products
Manufacturing documentation and traceability
- Managing batch records and ensuring accurate documentation of manufacturing processes
- Ensuring traceability of investigational products from production to trial sites
- Auditing manufacturing documentation for compliance with regulatory standards
- Coordinating with manufacturing teams to ensure timely documentation updates
- Maintaining clear communication with trial sites regarding product documentation
Quality control and assurance in manufacturing
- Implementing quality control procedures for clinical trial materials
- Conducting inspections of manufacturing sites for compliance with quality standards
- Managing deviations and non-conformances in the manufacturing process
- Collaborating with quality assurance teams to resolve manufacturing issues
- Ensuring that trial materials meet quality specifications before distribution
Regulatory compliance and GMP alignment
- Ensuring that manufacturing processes comply with GMP regulations
- Coordinating with regulatory affairs to align manufacturing practices with clinical trial requirements
- Preparing for and participating in regulatory inspections of manufacturing sites
- Implementing corrective actions based on regulatory audit findings
- Maintaining updated knowledge of GMP and regulatory requirements affecting clinical trials
Benefits Copied
Proficient personnel allocation through skills management software streamlines clinical trial coordination, optimizing recruitment, data collection, and protocol adherence.
Download the free Excel Clinical trial coordination skills matrix template Copied
We also have a free Excel template available that you can download if you are not ready to get started with AG5. To download it, please complete this form here.
Author Copied
Revisions Copied
Tired of managing skills in Excel?
Say goodbye to Excel matrices. Start using AG5’s plug and play skill matrix software.
Recognized by G2 for Excellence in Skills Management